Engaging outside experts to review risk assessments and methods can help insure high quality and scientifically-defensible work products and results. Government agencies, NGOs and industry recognize the value added by expert peer review and TERA is a world leader in providing independent expert review for all types of risk assessment documents and activities. TERA provides a variety of opportunities and services to engage expert peers, including in-person panel meetings, webcasts and webinars; letter reviews; workshops to develop risk values or methods; and in-house technical reviews.
Project Spotlight:
Independent Workshop on Ozone NAAQS Science and Policy
The U.S. Environmental Protection Agency (EPA) is proposing to lower the National Ambient Air Quality Standard (NAAQS) for ozone. TCEQ will host a 2.5 day public workshop to provide an independent evaluation and synthesis of key considerations for approaching the ozone NAAQS decision.
Project Spotlight:
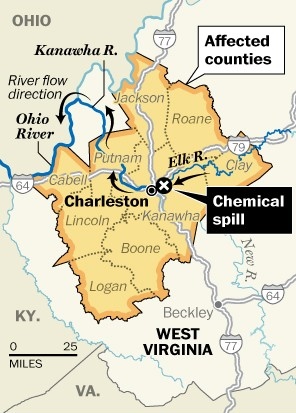
West Virginia Spill Peer Review - WV TAP Peer Review of the MCHM Screening Level
TERA has selected and convened a panel of five experts to review and discuss the available toxicology data and the scientific support for the West Virginia Screening Level established at 10 parts per billion (ppb). The panel discussed the initial starting value of 1 part per million (1,000 ppb) established by the US CDC and then considered if the additional safety factor applied by the State of West Virginia was protective.
Link to Workshop Page
Involvement of expert peers in the development and review of risk assessment documents and methodologies has expanded in recent years with government agencies and others seeking the benefits of having experts review work products before finalization. TERA has spent the last 10 years developing and refining procedures for involving experts in the development and review of risk assessment documents and methods. Through a variety of peer involvement tools, including peer input, peer consultation and peer review, TERA assists sponsors and authors in developing high quality results that are based on the best science. By engaging a broad range of experts from various backgrounds and perspectives, the assessment, method, protocol or document is strengthened through the identification of additional data and approaches, testing of assumptions, discussion of uncertainties, and shortcomings in logic or scientific rationales. Risk assessment is a multi-disciplined and complex process; no single person or organization has all the definitive answers for every situation. In many cases, involving others can strengthen the end result.
As a non-profit organization, TERA provides a variety of opportunities and services to engage expert peers in the development and improvement of risk assessment methods and documents. TERA’s work organizing peer input, peer consultation, and peer review is intended to meet the needs of public and private sponsors who have developed risk assessment documentation. Work products have included chemical assessments, hazard evaluations, site assessments, risk methodologies and guidance documents, protocols for studies, and research plans.
TERA works with each sponsor to design a peer involvement strategy that is most effective for the type of work product and stage of development. Options include peer input, peer consultation and peer review. We can plan open public meetings, letter reviews by mail, workshops, or other means of involving experts. We can engage external experts or utilize TERA scientists’ extensive experience in risk assessment, to provide the sponsor with the type of input that would be most helpful. Because the ultimate purpose of peer involvement is to help the authors and sponsors make sure their documents reflect the best science and analysis, we do not conduct the reviews in a vacuum. For most panel meetings, authors present their work and answer panel experts’ questions so that the experts may understand what the authors did and the authors may understand the experts comments and recommendations.
Our peer involvement efforts follow the principles that are the cornerstone of our program – scientific robustness, selection of appropriate expertise, and transparency. A fourth principle, independence, is key for all peer reviews and important for many peer input and consultation efforts. TERA conducts its scientific peer reviews in accordance with U.S. EPA peer review procedures (as described in EPA 2006). In addition, TERA has developed its peer review and consultation program following principles highlighted by the American Industrial Health Council’s 1995 report Fundamental Scientific Peer Review Principals and utilizing approaches used by the National Academy of Sciences and EPA’s Science Advisory Board. Peer input and peer consultations utilize the peer review guidance as appropriate.
For more information on the Independent Peer Review and Consultation Program, click on the links on the side bar or contact Dr. Patricia McGinnis